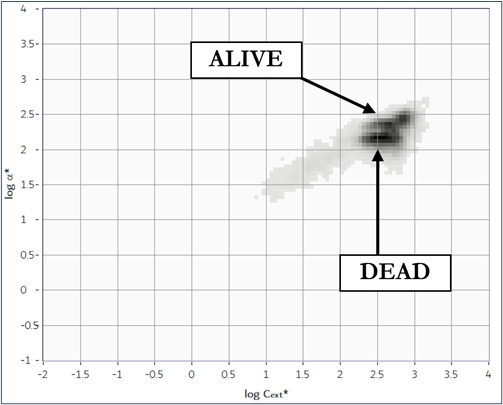
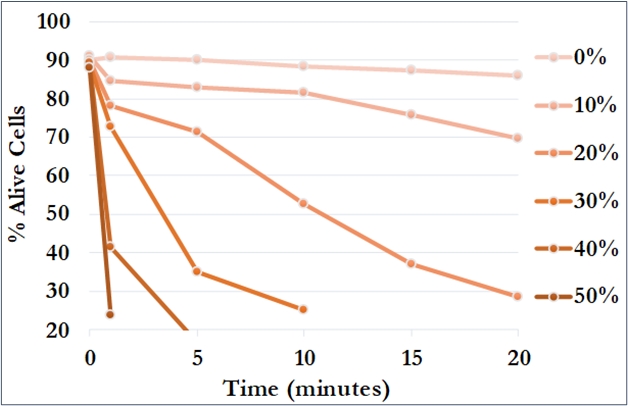
The cell viability is defined as a percentage of live cells in a whole population. The determination of yeast cell viability is commonly used to assessing the impact of various types of stressors in toxicity research and in industrial microbiology studies. The yeast Saccharomyces cerevisiae is a very useful model organism used for studies of cyto- or genotoxicity under different kinds of chemical, physical, or environmental factors. The analysis of the cell viability is also very important for industrial processes where microorganisms are used, as food, beverage, and biofuel. The Classizer™ ONE provides an accurate estimation of the cell viability regardless the presence of secondary populations and impurities.
In comparison with current fluorescence kit available on the market, SPES has mayor advantages:
- automatically reliable measure of ten thousand counts is performed.
- not time consuming: each single measure of this application note lasts less than few minutes.
- no expensive consumables.
- Iimited or even no sample treatment, except for a simple and automatable pre-dilution.
- inline/online monitoring in an industrial processes.